By 2030, multiple space agencies will have sent astronauts to the Moon for the first time since the Apollo Program ended over 50 years ago. These programs will create lasting infrastructure, like the Lunar Gateway, Artemis Base Camp, Moon Village, and the International Lunar Research Station (ILRS). In the ensuing decade, the first crewed missions to Mars are expected to occur, culminating with the creation of the first human outposts on another planet. Commercial ventures also want to establish habitats in Low Earth Orbit (LEO), enabling everything from asteroid mining to space tourism.
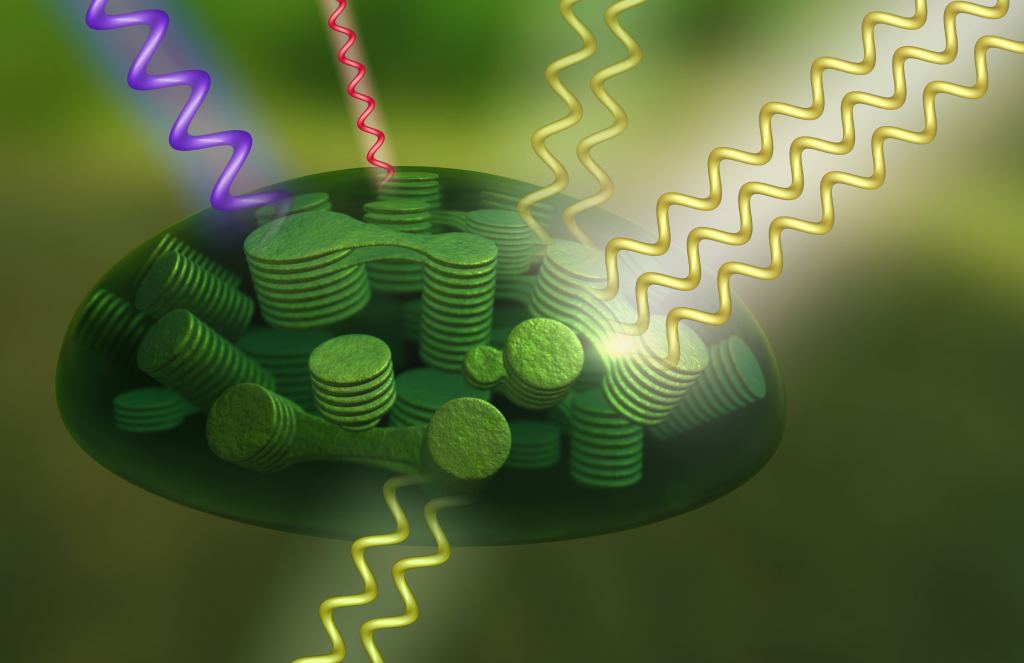
One of the biggest challenges for this renewed era of space exploration (Space Age 2.0) is ensuring that humans can remain healthy while spending extended periods in space. Foremost among them is ensuring that crews have functioning life support systems that can provide a steady supply of breathable air, which poses its own technical challenges. In a recent study, a team of researchers led by Katharina Brinkert of the University of Warwick described how artificial photosynthesis could lead to a new type of life support system that is smaller, lighter, easier, and more cost-effective to send to space.
In addition to being an assistant professor in Catalysis at the University of Warwick (UoW), UK, Dr. Brinkert is a researcher with the Center for Applied Space Technology and Microgravity (ZARM) at the University of Bremen, Germany. She was joined by Byron Ross, a Ph.D. student with Dr. Brinkert's research group (who led the study) in the Department of Chemistry at UoW, and Sophia Haussener, an associate professor with the Institute of Mechanical Engineering at Ecole Polytechnique Fédérale de Lausanne (EPFL). A paper that describes their latest research appeared on June 6th in *Nature Communications*.
Roughly 4 billion years ago, Earth's atmosphere and environment were much different than they are today. Known as the Archean Eon, the planet was covered with active volcanoes, and its atmosphere was predominantly composed of carbon dioxide (CO2), sulfur dioxide, and other volcanic gases. During this same period, the first lifeforms are thought to have emerged: single-celled microbes that relied on retinol or chlorophyll for photosynthesis. These lifeforms slowly converted the atmosphere, combining water, sunlight, and CO2to create glucose (an energy source) and oxygen gas as a byproduct.
This led to the " Great Oxidation Event " and the emergence of more complex life forms, eventually giving rise to hominids and homo sapiens (aka. humanity). So integral is photosynthesis to life on Earth that scientists hope to harness the process to facilitate living in space. On the International Space Station (ISS), astronauts rely on the Environmental Control and Life Support System (ECLSS) to provide a steady oxygen supply. This system relies on electrolysis, where electricity from the station's solar panels splits water into hydrogen and oxygen gas.
Meanwhile, a separate system "scrubs" carbon dioxide from the air and converts it into water and methane. Unfortunately, these systems are large, bulky, difficult to maintain, and inefficient, requiring about one-third of the energy needed to power the ECLSS. Replenishing the system is relatively easy since missions in Low Earth Orbit (LEO) can be resupplied in hours. But for missions headed to Mars, which can spend six to nine months in transit and take up to three years, resupply missions are not practical. As Dr. Brinkert related in a recent op-ed with The Conversation:
"The search for alternative systems which can be employed on the Moon and on trips to Mars is therefore ongoing. One possibility is to harvest solar energy (which is abundant in space) and directly use it for oxygen production and carbon dioxide recycling in only one device. The only other input in such a device would be water - similar to the photosynthesis process going on in nature. That would circumvent complex set-ups where the two processes of light harvesting and chemical production are separated, such as on the ISS."
Instead of chlorophyll, which plants and algae rely on to absorb sunlight, a photoelectrochemical (PEC) system would rely on semiconductor materials coasted with metallic catalysts to convert CO2 and water into oxygen gas and hydrogen/carbon-based fuels. Additional energy generated by solar heating could be harnessed to directly catalyze water and speed up the chemical process, thereby requiring less electricity. This life support system would be highly beneficial for long-duration missions since it would combine reduced volume and weight with greater efficiency.
Furthermore, the system would be easier to maintain since it would need less complex wiring and mechanical components. To test this concept, Dr. Brinkert and her colleagues produced a theoretical framework for measuring the performance of a PEC against a conventional BLSS. This included the efficiency of solar-driven water splitting, the available solar energy on Mars (roughly half of what Earth receives), and the reduction of CO2 in Mars' atmosphere. Lastly, they considered how solar concentrator devices could assist PEC devices and how they could be manufactured via in-situ resource utilization (ISRU). Said Blinkert:
"On Mars, the atmosphere [is composed] of nearly 96% carbon dioxide - seemingly ideal for an artificial photosynthesis device. But the light intensity on the Red Planet is weaker than on Earth due to the larger distance from the Sun. So would this pose a problem? We actually calculated the sunlight intensity available on Mars. We showed that we can indeed use these devices there, although solar mirrors become even more important.
"Our analysis shows that these devices would indeed be viable to complement existing life support technologies, such as the oxygen generator assembly employed on the ISS. This is particularly the case when combined with devices that concentrate solar energy in order to power the reactions (essentially large mirrors which focus the incoming sunlight)."
This proposed system offers numerous advantages over conventional electrolysis devices, which operate at high temperatures and require a lot of electricity. While NASA is investigating technology that could harvest oxygen directly from lunar regolith, this method requires very high temperatures to convert elemental oxygen into oxygen gas (O2). On the other hand, PEC devices could operate at room temperature within Martian and lunar habitats, using water as the main resource. The abundance of water ice on Mars and in the cratered South Pole-Aitken Basin makes this method particularly attractive.
The technology is still in its infancy and requires several more years of additional research, says Blinkert. But the potential returns would be immense, ranging from lightweight life support systems for long-duration missions to artificial atmospheres for habitats in LEO, on the Moon, and on Mars. But of course, the benefits would go beyond space exploration and could have applications here at home. Said Blinkert:
"[T]he insights we gain from designing and fabricating these devices could help us meet the green energy challenge on Earth. We are fortunate enough to have plants and algae for producing oxygen. But artificial photosynthesis devices could be used to produce hydrogen or carbon-based fuels (instead of sugars), opening up a green way for the production of energy-rich chemicals which we can store and use in transport. The exploration of space and our future energy economy have a very similar long-term goal: sustainability. Artificial photosynthesis devices may well become a key part of realizing it."
This research mirrors similar efforts to develop life support systems that mimic biological systems here on Earth. These proposed bioregenerative life support systems (BLSS) could replenish themselves over time, providing oxygen, water, and even food in a way that is sustainable. The development of this technology will ensure humanity's future in space and allow for more sustainable living here on Earth, helping to mitigate the effects of Climate Change.
*Further Reading: The Conversation*, Nature Communications
 Universe Today
Universe Today