[/caption]
Yep. It's true. Almost all galaxies are guilty of having a supermassive black hole in their centers. Some even tip the scales at millions - or even billions - of times more mass than the Sun. However, how they came to be so weighty is a true enigma. Thanks to research done by Dr. John Silverman (IPMU) and the international COSMOS team, the Chandra X-Ray Observatory and the European Southern Observatory's Very Large Telescope have revealed that galaxy interactions may be responsible for the growth of supermassive black holes - and they've left behind some very important clues...
If you're big - you're big. As a general rule, supermassive black holes like to hang out in massive galaxies. Their mass is usually directly related to the central bulge. Now the consensus is that massive galaxies gained their girth (at least in part) by mergers and interactions with smaller galaxies. This act of cannibalism in galactic evolution has been postulated to explain how matter gathers toward the middle, eventually resulting in a supermassive black hole.
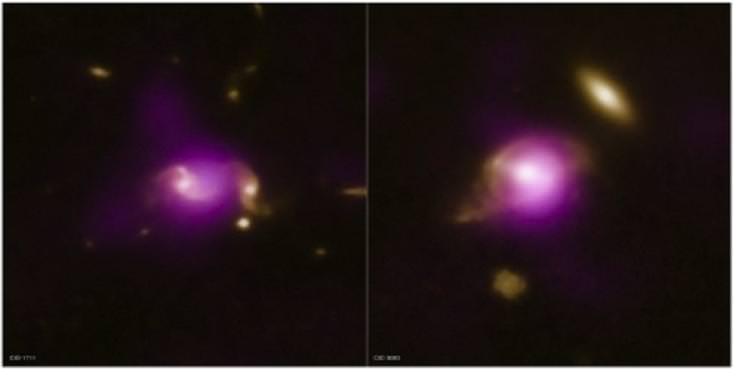
How do we determine this? One way is to take a closer look at galaxies currently in merger as compared to ones in isolation. While the concept is easy, carrying out the test hasn't been. A supermassive black hole leaves visual observations "blinded by the light" while a quasar can effectively "outshine" an entire host galaxy, leaving an interactor almost impossible to detect. But, like a bulging waistline, such interactions should distort the overall contours of the galaxy.
Now the COSMOS team might have an answer to the riddle.. by assuming a galaxy is interacting if it has a nearby neighbor. It's a test that can happen without needing to know if distortion is present in optical images. What makes it possible are accurate distance measurements of about 20,000 galaxies in the COSMOS field as provided by the zCOSMOS redshift survey with the European Southern Observatory's Very Large Telescope. Isolated galaxies are used to give a comparison sample to lay the foundation as to whether an active galactic nucleus is common to interacting galaxies. With help from NASA's Chandra Observatory, X-ray observations pinpoint galaxies which host an AGN. The X-ray emission signature dominates in growing SMBHs and X-rays are capable of cutting through the gas and dust of star-forming regions.
In their report to
The Astrophysical Journal
the team states that galaxies in close pairs are twice as likely to harbor AGNs as compared to galaxies in isolation. This answer may prove that beginning galaxy interactions can lead to "enhanced black hole growth". Because it's not a drastically common occcurrance, it means that only about 20% of SMBHs that break the scale happen via a merger event and that "final coalescence" might also play a role.
One thing we do know is that galaxies and their black holes, like people and their waistlines, all get a little heavier with time.
Original Story Source: Institute for Physics and Mathematics of the Univserse.
 Universe Today
Universe Today