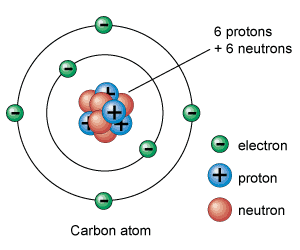
[/caption]
Ever wonder why the periodic table of elements is organized the way it is? Why, for example, does Hydrogen come first? And just what are these numbers that are used to sort them all? They are known as the element's atomic number, and in the periodic table of elements, the atomic number of an element is the same as the number of protons contained within its nucleus. For example, Hydrogen atoms, which have one proton in their nucleuses, are given an atomic number of one. All carbon atoms contain six protons and therefore have an atomic number of 6. Oxygen atoms contain 8 protons and have an atomic number of 8, and so on. The atomic number of an element never changes, meaning that the number of protons in the nucleus of every atom in an element is always the same.
Arranging elements based on their atomic weight began with Ernest Rutherford in 1911. It was he who first suggested the model for an atom where the majority of its mass and positive charge was contained in a core. This central charge would be roughly equal to half of the atoms total atomic weight. Antonius van den Broek added to this by formerly suggesting that the central charge and number of electrons were equal. Two years later, Henry Moseley and Niels Bohr made further contributions that helped to confirm this. The Bohr model of the atom had the central charge contained in its core, with its electrons circulating it in orbit, much like how the planet in the solar system orbit the sun. Moseley was able to confirm these two hypotheses through experimentation, measuring the wavelengths of photon transitions of various elements while they were inside an x-ray tube. Working with elements from aluminum (which has an atomic number thirteen) to gold (seventy nine), he was able to show that the frequency of these transitions increased with each element studied.
In short, the higher the atomic number (aka. the higher the number of protons), the heavier the element is and the lower it appears on the periodic table. The atomic number of an element is conventionally represented by the symbol Z in physics and chemistry. This is presumably derived from the German word Atomzahl, which means atomic number in English. It is not to be confused with the mass number, which is represented by A. This corresponds to the combined mass of protons and neutrons in the element.
We have written many articles about the atomic number for Universe Today. Here's an article about the
atomic nucleus
, and here's an article about the
Atom Models
.
If you'd like more info on the Atomic Number, check out
NASA's Atoms and Light Energy Page
, and here's a link to
NASA's Atomic Numbers and Multiplying Factors Page
.
We've also recorded an entire episode of Astronomy Cast all about the Atom. Listen here,
Episode 164: Inside the Atom
.
Sources:
NDT Resource Center
Jefferson Lab
Wise Geek
Wiki Answers
 Universe Today
Universe Today