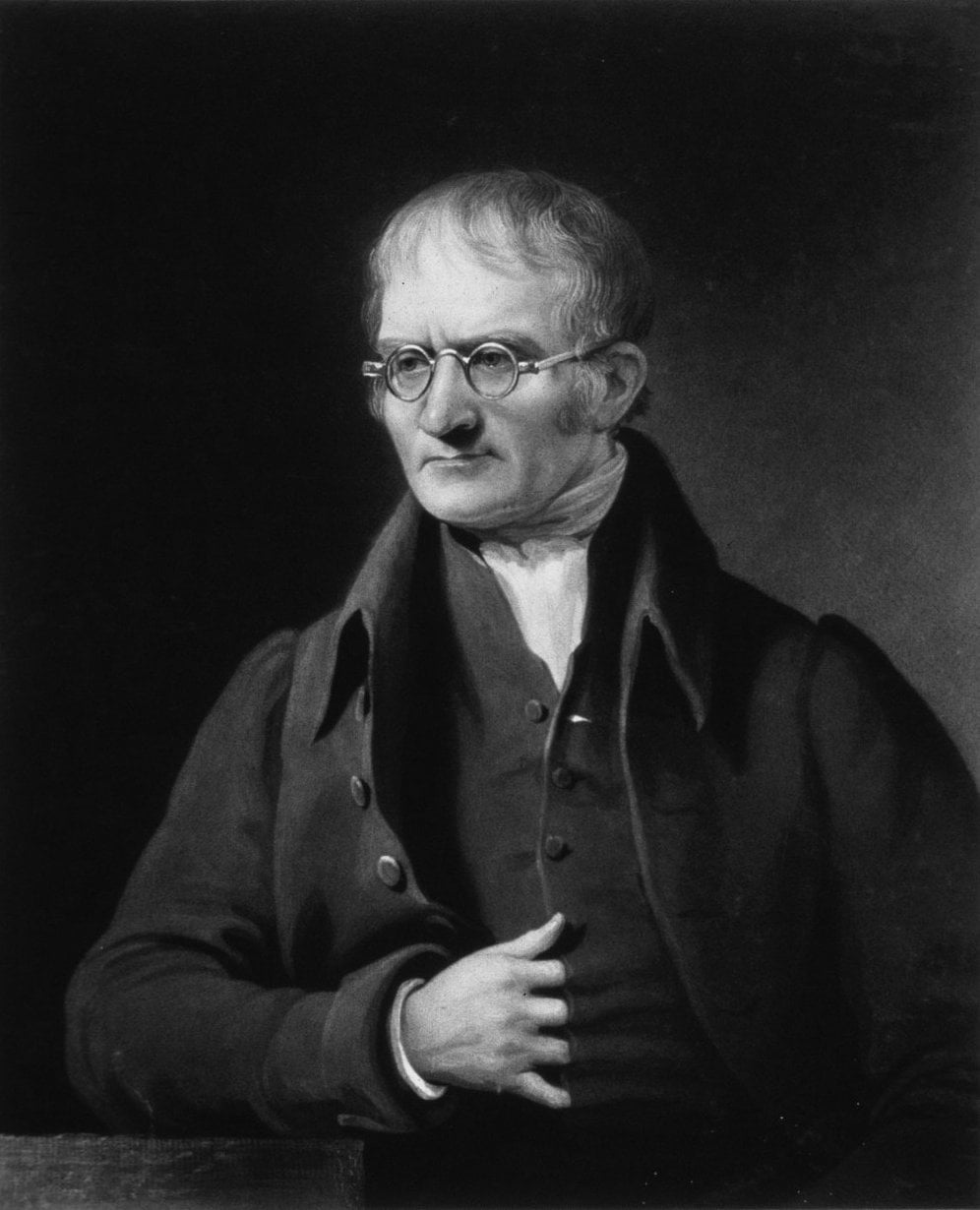Atomic theory - that is, the belief that all matter is composed of tiny, indivisible elements - has very deep roots. Initially, the theory appeared in thousands of years ago in Greek and Indian texts as a philosophical idea. However, it was not embraced scientifically until the 19th century, when an evidence-based approach began to reveal what the atomic model looked like.
It was at this time that John Dalton, an English chemist, meteorologist and physicist, began a series of experiments which would culminate in him proposing the theory of atomic compositions - which thereafter would be known as Dalton's Atomic Theory - that would become one of the cornerstones of modern physics and chemistry.
Beyond creating a model for atomic interactions, John Dalton is also credited with developing laws for understanding how gases work. In time, this would lead him to conclude things about how atoms interacted, the weight of atoms, and to design laws that establish atomic theory as scientific discipline.
Dalton's Gas Laws:
Dalton came up with his theory of atoms as a result of his research into gases. This began in 1800, when Dalton became a secretary of the Manchester Literary and Philosophical Society. While there, Dalton began submitted a series of essays that outlined his experiments on the constitution of mixed gases, including the pressure of steam and other vapors at different temperatures, on evaporation. and on the thermal expansion of gases.
In his essays, Dalton described experiments in which he sought to ascertain the pressure of steam at various points between 0 and 100 °C (32 and 212 °F). Based on his observations of six different liquids, Dalton concluded that the variation of vapor pressure for all liquids was equivalent, for the same variation of temperature, and the same vapor of any given pressure.
He also concluded that all elastic fluids under the same pressure expand equally when heat is applied. Further, he observed that for any given expansion of mercury (i.e. noted rise in temperature using a mercury thermometer), that the corresponding expansion of air is proportionally less, the higher the temperature goes.
[caption id="attachment_116699" align="aligncenter" width="578" class=" "]
A depiction of the atomic structure of the helium atom. Credit: Wikipedia/Creative Commons
[/caption]
This became the basis as Dalton's Law (aka. Dalton's law of partial pressures), which stated that in a mixture of non-reacting gases, the total pressure exerted is equal to the sum of the partial pressures of the individual gases.
Dalton's Atomic Theory:
In the course of this research into gases, Dalton also discovered that certain gases could only be combined in certain proportions, even if two different compounds shared the same common element or group of elements.
These experiments built upon two theories that had emerged near the end of the 18th century which dealt with chemical reactions. The first was the law of
conservation of mass
, formulated by Antoine Lavoisier in 1789, which states that the total mass in a chemical reaction remains constant - i.e. that the reactants have the same mass as the products.
The second was the
law of definite proportions
, first proven by the French chemist Joseph Louis Proust in 1799. This law states that if a compound is broken down into its constituent elements, then the masses of the constituents will always have the same proportions, regardless of the quantity or source of the original substance.
[caption id="attachment_116698" align="aligncenter" width="580" class=" "]
Various atoms and molecules as depicted in John Dalton's A New System of Chemical Philosophy (1808). Credit: Public Domain
[/caption]
Studying these laws and building on them, Dalton developed his
law of multiple proportions
. This law states that if two elements can be combined to form a number of possible compounds, then the ratios of the masses of the second element, which combine with a fixed mass of the first element, will be ratios of small whole numbers.
In other words, elements combine at the atomic level in fixed ratios which naturally differ based on the compounds being combined, due to their unique atomic weights. The findings became the basis of of Dalton's Atomic Laws or Model, which focuses on five basic theorems. T
The state that elements, in their purest state, consist of particles called atoms; that atoms of a specific element are all the same, down to the very last atom; that atoms of different elements can be told apart by their atomic weights; that atoms of elements unite to form chemical compounds; and that atoms can neither be created or destroyed in chemical reaction, only the grouping ever changes.
Dalton also believed atomic theory could explain why water absorbed different gases in different proportions - for example, he found that water absorbed carbon dioxide far better than it absorbed nitrogen. Dalton hypothesized this was due to the differences in mass and complexity of the gases' respective particles.
[caption id="attachment_126938" align="aligncenter" width="570"]
Dalton's model of the atom, based on the five points of his atomic theory. Credit: modernbiochemistry.weebly.com
[/caption]
In fact, it was this very observation that is believed to be the first time that Dalton hinted at the supposed existence of atoms. In the paper which addressed gas absorption in water, which was first published in 1805, he wrote:
"Why does not water admit its bulk of every kind of gas alike? This question I have duly considered, and though I am not able to satisfy myself completely I am nearly persuaded that the circumstance depends on the weight and number of the ultimate particles of the several gases
."
Dalton proposed that each chemical element is composed of atoms of a single, unique type, and though they cannot be altered or destroyed by chemical means, they can combine to form more complex structures (i.e. chemical compounds). This marked the first truly scientific theory of the atom, since Dalton reached his conclusions by experimentation and examination of the results in an empirical fashion.
Dalton And Atomic Weights:
Dalton's also began studying atomic weights based on the mass ratios in which they combined, with the hydrogen atom taken as the standard. However, Dalton was limited by the crudity of his laboratory instruments and the fact that he did not conceive that the atoms of certain elements exist in molecular form, such as pure oxygen (O
2
).
He also believed that the simplest compound between any two elements is always one atom each. This was best illustrated in how he thought that the chemical formula for water was HO, not H
2
O.
[caption id="attachment_126941" align="aligncenter" width="553"]
John Dalton, the father of modern atomic theory. Credit: chemheritage.org
[/caption]
In 1803, Dalton orally presented his first list of relative atomic weights for a number of substances. This paper was published in 1805, but he did not discuss there exactly how he obtained these figures. In 1807, his method was revealed by his acquaintance Thomas Thomson, in the third edition of Thomson's textbook,
A System of Chemistry
. Finally, Dalton published a full account in his own textbook,
A New System of Chemical Philosophy
, in 1808 and 1810.
Scientific Flaws:
The main flaw in Dalton's theory - i.e. the existence of both molecules and atoms - was later corrected in principle in 1811 by Amedeo Avogadro. Avogadro proposed that equal volumes of any two gases, at equal temperature and pressure, contain equal numbers of molecules. In other words, the mass of a gas's particles does not affect the volume that it occupies.
Avogadro's law allowed him to deduce the diatomic nature of numerous gases by studying the volumes at which they reacted. Thus, Avogadro was able to offer more accurate estimates of the atomic mass of oxygen and various other elements, and made a clear distinction between molecules and atoms. Alas, these and other discoveries both contradicted and refined Dalton's theories.
For instance, scientists have since discovered that the atom - once thought to be the smallest part of matter - can in fact be subdivided into even smaller elementary particles. And whereas Dalton conceived as atoms as a single entity with no separation between positive, negative, and neutral charges, subsequent experiments by
J.J. Thomson
, Ernest Rutherford, and
Neils Bohr
revealed a more complex structure to the atom.
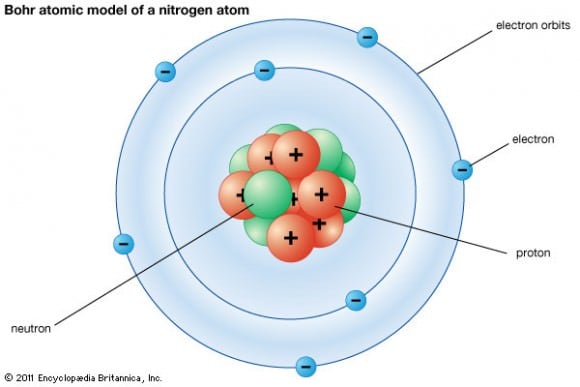
[caption id="attachment_123893" align="aligncenter" width="580"]
Neils Bohr's model a nitrogen atom. Credit: britannica.com
[/caption]
These theories were later validated by observations made with the electron microscope. We also know that atomic weight is a product of the structure of the atoms themselves. Hence, Dalton's Atomic Model, in its purest form, is now only considered valid for chemical reactions. However, this does not diminish Dalton's contribution to modern science.
Prior to his time, the atom was little more than a philosophical construct passed down from classical antiquity. Dalton's groundbreaking work not only made the theory a reality but led to many other discoveries, such as
Einstein's theory of relativity
and Planck's quantum theory - two fields of study that form the basis of our modern understanding of the cosmos.
We have written many interesting articles about atomic theory here at Universe today. Here is one about the number of
Atoms In The Universe
,
What Are The Parts Of The Atom?
,
Who Was Democritus?
,
Bohr's Atomic Model
, and
What Is The Plum Pudding Model?
If you want to learn more about Dalton's model, check out the article from Central Queensland University about
Dalton's Atomic model
.
Astronomy Cast has recorded many interesting episodes on the subject. Check them out -
Episode 138: Quantum Mechanics
,
Episode 378: Rutherford and Atoms
, and
Episode 392: The Standard Model - Intro
.
 Universe Today
Universe Today