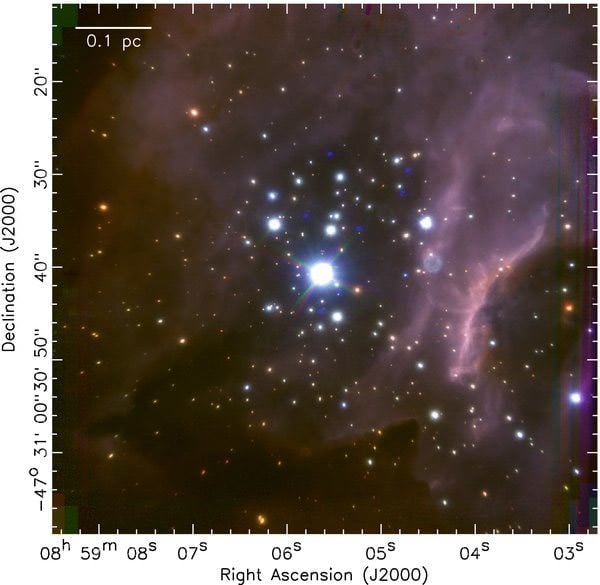
[/caption] Stellar nurseries can be found in giant clouds of molecular gas and dust scattered throughout our galaxy. The Octo-mom has nothing on these stellar nurseries, as these regions can produce multiple stars at once – into the hundreds at a time. How often does this happen? On average, one new star is born somewhere in our Milky Way galaxy per year, astronomers estimate. But with the newborns arriving together in dense clusters, stars aren't born, or created, very often in the Milky Way. Recently, astronomers took a close look in infrared at what was happening inside a giant stellar nursery called RCW 38 and saw hundreds of stars in different stages of development. What they found was significant, as this represents the first time a massive cluster other than the one in the Orion Nebula has been studied so precisely.
RCW 38 is located about six thousand light-years away, and is one of only two relatively nearby giant clusters with over 1000 stars. The other one is the Orion Nebula, which is 3.5 times closer and much easier to study, and so thus far a unique example.
The astronomers studies 317 stars in the cluster at three infrared wavelengths. About thirty percent of them were noticeably red, suggesting the presence of circumstellar, perhaps protoplanetary, disks. They also found traces of shocked gas and a few even younger protostars, all features consistent with this being an active stellar nursery.
This initial study is expected to be followed up with more in-depth looks to determine which features of a cluster are characteristic of all clusters, and which (for example the spatial distribution of the stars, the numbers of different kinds of stars, or the numbers of stars with protostellar disks) are only circumstantial.
Future studies will also tell us more about our own solar system. One line of thinking is that our sun may have formed in a cluster that later dissipated. Since ultraviolet light can evaporate dust, massive hot stars that emit such light may have played a role by inhibiting the formation of planets if they were near the young sun; likewise, if a nearby massive star exploded as a supernova during the early days of the sun, the event might explain the abundances of radioactive elements found in the solar system.
Read more about this study of RCW 38 in the team's paper.
 Universe Today
Universe Today