*Author's note: This article was written in collaboration with Vincent Kofman, a co-author of the paper it discusses and Post Doctoral Researcher at NASA's Goddard Space Flight Center*
Amino acids are one of the most important building blocks of life as we know it. At its core, they contain an amino and an acid group, through which they can link together with other amino acids. That linking process can form long chains, which is how they form proteins. In humans, 20 different amino acids make up all proteins, and the difference between them is in the molecular side chain between the amino and the acid group. The different groups make interconnections in the chain, folding it into highly specific forms, allowing the proteins to perform highly specific tasks, ranging from metabolism, to muscle movement, and cell duplication.
Given that their presence is a necessary, though not necessarily a sufficient, condition for the development of life, scientists have spent many decades exploring where they first formed. With a paper in Nature Astronomy published last month, they moved one step closer to that understanding, by discovering that it is possible to form glycine, the simplest amino acid, in the star nurseries of interstellar clouds.
Glycine's development in such an inhospitable environment had previously been theorized. Most notably, glycine has been observed in samples of comet Wild 2 returned from the Stardust mission as well as in observations of the coma of comet 67P/Churymov-Gerasimenko. Since comets formed together with the solar system from the same original starting material, they are a window into that solar system in its earlier years. Glycine has been seen in those windows, so scientists theorized that it might have formed in the base material that both comets and the solar system itself share.
A team led by Sergio Ioppolo of Queen Mary University set out to see if they could form glycine in the conditions of the interstellar gas clouds where stars, planets, and comets are born.. Their work mirrored another famous chemistry experiment searching for the molecules required for the origins of life - the Miller-Urey experiment. In that work, scientists tried to recreate conditions thought to be present on the early Earth by throwing a "primordial soup" of chemicals together and added lightning to the mix. Under these conditions, Miller and Urey found a heap of organic molecules which could be the of precursors to the first sparks of life.
Dr. Ioppolo and his team tried to work out the same question, but looked very specifically at how these essential molecules of life are formed. They constructed an ultra-high vacuum chamber, lowered the temperature down to between 10-20 degrees Kelvin to mimic the conditions in dark interstellar gas clouds. Subsequently, they placed chemicals known to be present in those interstellar clouds inside the vacuum chamber, such as carbon monoxide, ammonia, and methane. Then they exposed the frozen chemicals to unbound atoms, that sparked chemistry similar to that which takes place under dark cloud conditions. Like magnets clicking together, the atoms broke apart the existing chemicals and allowed their constituents to recombine in unique ways.
The chemical conditions in dark clouds are very different from those here on the Earth. The density of particles is millions of billions of times less, and the temperatures are so low that almost everything freezes solid. Under these conditions, molecules form atom by atom, and like bricks of Lego, specific molecules can be built up from scratch. By recreating these conditions in the laboratory, scientists can figure out what chemical reactions are likely to take place, and which molecules may form.
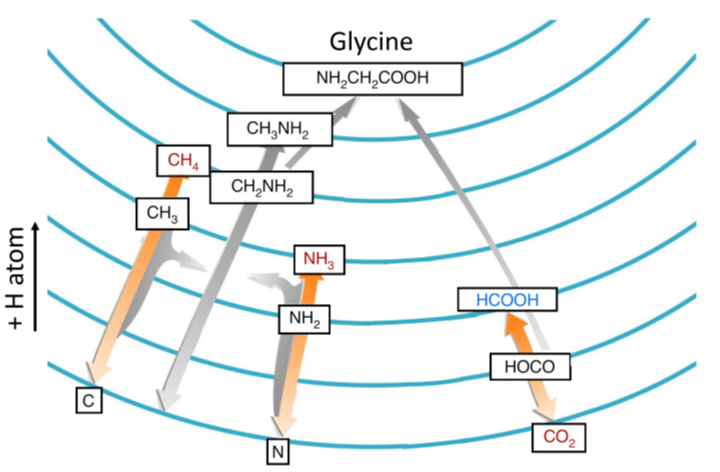
The work by Dr. Ioppolo and his team did exactly that, and demonstrated that glycine can form under the conditions that occur in these dark corners of space. Similar to the Sun, most of the material in dark clouds consists of helium and hydrogen. Hydrogen, can exist as a molecule, H2, and as an unbound atom.
The unbound atom is highly reactive, and initiates chemistry with almost anything it comes across. It combines with other atoms build up molecules such as CH4 (methane), NH3 (ammonia). In the intermediate stages of this hydrogen bombardment, the precursor chemicals CH3 and NH2 may react to form CH3NH2, methylamine. Methylamine is a precursor to glycine and has been found abundantly in comet 67P. This finding most likely directly leads to the finding of the abundance of glycine in the same comet.
In addition to creating methane or amonia, hydrogen could also react with CO2, which is also abundantly present in the icy layers in the dark clouds. When it does so, it enables the carbon atom to from another bond. Sometimes that additional bond will form to methylamine, which results in glycine. In experiments described in the recent paper, the specifics of these reactions were investigated, and the abundance of glycine formed in these reactions was calculated. The calculation shows that the amount of glycine that is formed is enough to enrich planets like the early Earth with prebiotic molecules to facilitate the origins of life.
Now that the chemical proof of concept has been shown, scientists can turn to the numerous implications this work has for both existing theories and future studies. The paper shows that it is possible that the building blocks of life can result from natural processes taking place before star formation, enriching the raw materials of planets with the essential building blocks of life. Although these molecules may not survive the harsh events of planet formation, the fact that they may be present in the raw materials of stars and planets suggests their abundance is widespread throughout the universe.
Although only glycine had been found in the experiments reported in the paper, demonstrating the formation of the amino acid backbone opens the pathway for the formation of other amino acids as well. Earlier research has shown the central carbon atom, which carries the group that gives amino acids their function, is most likely to be targeted in reactions taking place in the same ices as they are formed. This indicates that the other biologically relevant amino acids may also form under the same conditions.
The study also leads to some additional intriguing questions. If the glycine is present, how can we directly detect it? Under the right circumstances, telescopes may be able to detect glycine if it is heated up, and brought into the gas phase. When new stars start heating up the surrounding material, the primordial ices are melted, and the constituents can be detected using ground-based observatories such as the Atacama Large Millimeter Array (ALMA) in Chile. Peering deep into these stellar nurseries has revealed a complex chemistry and taught us much about star and planet formation.
If the building blocks of life are present in the raw materials of stars and planets, this naturally leads us to wonder what the chances are that life is present in other places in the Milky Way. If our Solar System alone contains a handful of planets and moons where life may be possible, we can only imagine how many planets and stars are out there that may harbor life. When put in an appropriate context, this better understanding of the formation of the building blocks of life truly has some staggering implications for the potential abundance of life in throughout the galaxy.
Learn More:
Nature - A non-energetic mechanism for glycine formation in the interstellar medium
Queen Mary University of London - Building blocks of life can form long before stars
Sci News - Amino Acids Can Form Long Before Stars and Planets: Study
C&EN - Interstellar Glycine Search Intensifies
PhysicsWorld - Amino Acid Detected In Space
Lead Image:
Artist's depiction of an environment the experimenters were going for to create glycine.
Credit: Professor Harold Linnartz
 Universe Today
Universe Today