The liver plays a vital role in human health, regulating metabolism and blood nutrient levels, filtering toxins, and synthesizing important proteins for blood clotting. It is also sensitive to changes in diet, behavior, and environmental factors, meaning it will also respond to changes in gravity. Despite considerable research into liver metabolism and function aboard the International Space Station (ISS), unanswered questions remain about how liver cells sense gravity and convert mechanical stress into metabolic responses.
While the research has suggested a strong link between microgravity and the accumulation of fatty cells in liver tissue, knowing how this process is triggered is key to developing mitigation strategies. To address this, a team of Chinese scientists investigated the effects of microgravity on two cultures of liver cells, one aboard the Tiangong space station and the other on Earth. Their results offer some potential solutions for monitoring and mitigating the effects of microgravity on the liver.
The team was led by Professor Mian Long from the Institute of Mechanics, Chinese Academy of Sciences. He was joined by researchers from the Center for Biomechanics and Bioengineering, the Beijing Key Laboratory of Engineered Construction and Mechanobiology, the Key Laboratory of Microgravity, the University of Chinese Academy of Sciences, and the Key Laboratory of Biorheological Science and Technology. Their work is detailed in a paper published in Science Bulletin.
 Understanding the effects of microgravity on liver health is paramount for astronauts on future deep-space missions. Credit: Li, N. et al. (2026)
Understanding the effects of microgravity on liver health is paramount for astronauts on future deep-space missions. Credit: Li, N. et al. (2026)
Research conducted aboard the ISS has consistently shown a link between microgravity and hepatic lipid metabolism, the process by which the liver regulates energy balance by synthesizing, breaking down, and transporting fats. This is characterized by neutral lipids accumulating within the primary tissue cells of the liver (hepatocytes), responsible for bile secretion, glucose regulation, lipid uptake, and urea production. Research has also shown that prolonged exposure to microgravity leads to an increased risk of metabolic dysfunction–associated steatotic liver disease (MASLD).
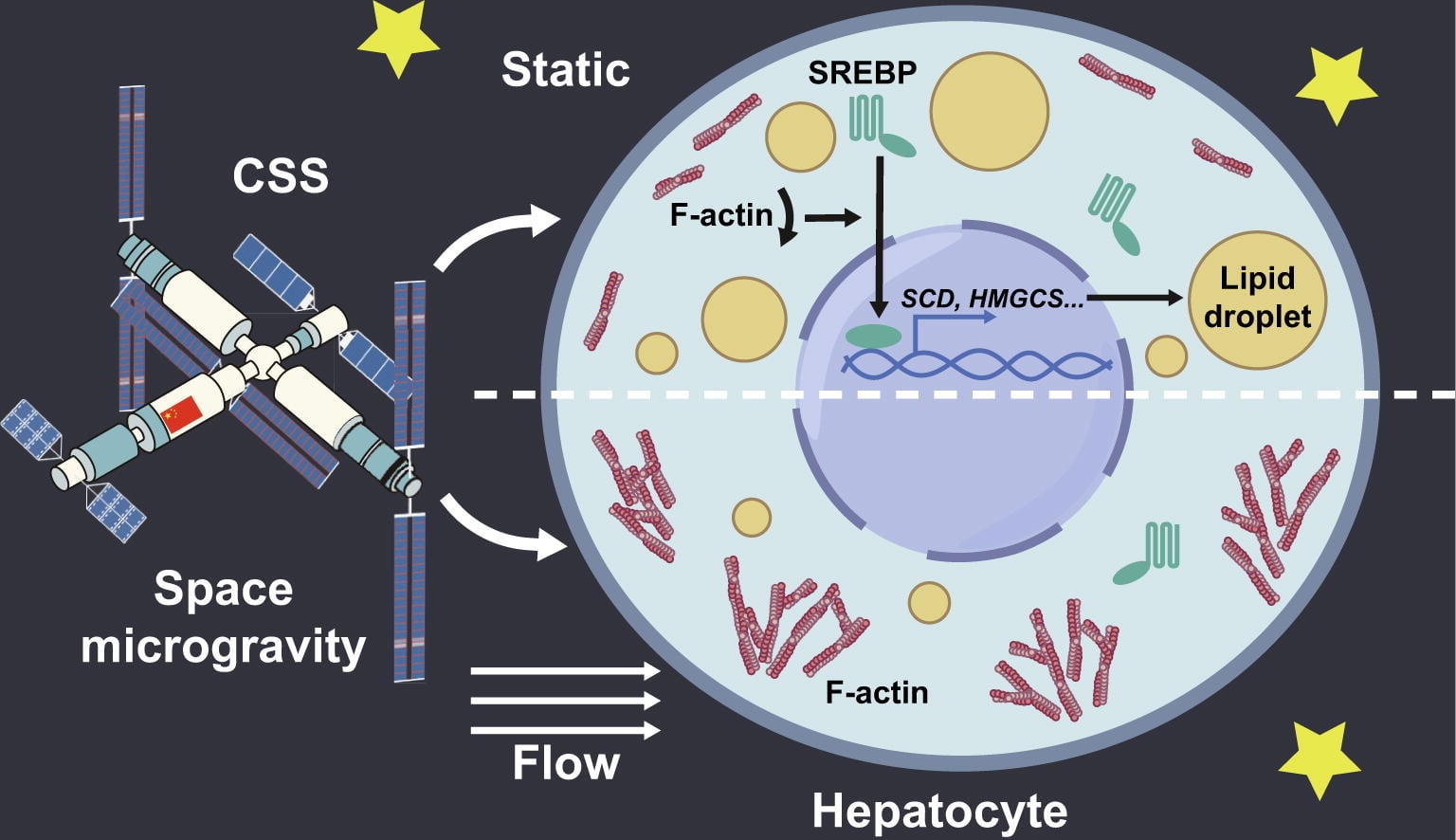
Increased lipid dysregulation and hepatic damage constitute major health risks for astronauts engaged in future deep-space exploration missions. To investigate the mechanics of spaceflight and liver function, the team cultured two hepatocyte samples in a Biomechanics Experiment Module (BMEM). One sample was sent to the Tiangong space station while the other remained on Earth. These samples were divided into three experimental groups based on culturing and exposure to shear flow stress.
This stress simulated the normal movement of fluid within the liver (i.e., blood flow), specifically in the area between hepatocytes and sinusoidal endothelial cells. The samples were then observed for nine days to monitor for any changes. The results showed that spaceflight promotes the synthesis of fatty acids and cholesterol by activating sterol regulatory element-binding proteins (SREBPs), effectively demonstrating that SREBPs could serve as gravity-sensitive regulators of lipid accumulation in hepatocytes.
The results further indicate that SREBPs have potential as therapeutic targets for maintaining liver health during long-duration space missions. In addition, analysis of the samples upon their return to Earth showed that shear flow exerted a protective effect, partially mitigating lipid dysregulation. In short, the tests demonstrated a means to monitor liver function in space while offering a mitigation strategy for future missions.
Further Reading: Science China Press, Science Bulletin
 Universe Today
Universe Today
