[/caption]
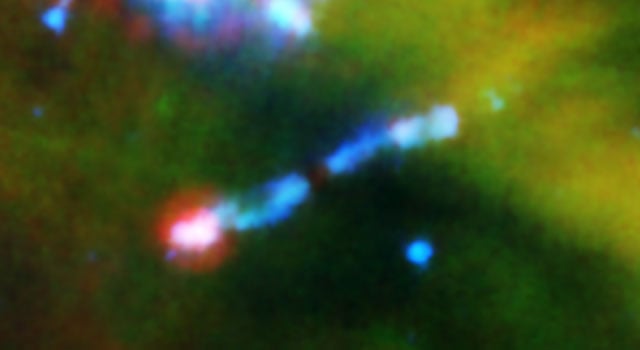
The Spitzer Space Telescope has spied water in a cloud of gas and dust around a nascent star. That's interesting in itself, but even more remarkable, the water is being blasted apart by the young star's laser-like jets. Spitzer's spectrometer was used to get a better look at these jets and analyze the jet's molecules. To the astronomers' surprise, Spitzer picked up the signature of rapidly spinning fragments of water molecules, called hydroxyl, or OH. "This is a truly unique observation that will provide important information about the chemistry occurring in planet-forming regions, and may give us insights into the chemical reactions that made water and even life possible in our own solar system," said Achim Tappe, of the Harvard-Smithsonian Center for Astrophysics, Cambridge, Mass.
A young star forms out of a thick, rotating cloud of gas and dust. Like the two ends of a spinning top, powerful jets of gas emerge from the top and bottom of the dusty cloud. As the cloud shrinks more and more under its own gravity, its star eventually ignites and the remaining dust and gas flatten into a pancake-like disk, from which planets will later form. By the time the star ignites and stops accumulating material from its cloud, the jets will have died out.
Tappe and his colleagues used Spitzer's infrared eyes to cut through the dust surrounding the star, called HH 211-mm, to analyze the jets. The astronomers were surprised to see water molecules in the data. But the results showed the hydroxyl molecules have absorbed so much energy (through a process called excitation) that they are rotating around with energies equivalent to 28,000 Kelvin (27,700 degrees Celsius). This far exceeds normal expectations for gas streaming out of a stellar jet. Water, which is abbreviated H2O, is made up of two hydrogen atoms and one oxygen; hydroxyl, or OH, contains one oxygen and one hydrogen atom.
The results reveal that the jet is ramming its head into a wall of material, vaporizing ice right off the dust grains it normally coats. The jet is hitting the material so fast and hard that a shock wave is also being produced.
"The shock from colliding atoms and molecules generates ultraviolet radiation, which will break up water molecules, leaving extremely hot hydroxyl molecules," said Tappe.
Tappe said this same process of ice being vaporized off dust occurs in our own solar system, when the sun vaporizes ice in approaching comets. In addition, the water that now coats our world is thought to have come from icy comets that vaporized as they rained down on a young Earth. This discovery provides a better understanding of how water -- an essential ingredient for life as we know it -- is processed in emerging solar systems.
Source:
JPL
 Universe Today
Universe Today