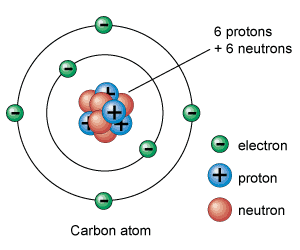
[/caption]The most widely accepted atom model is that of Niels Bohr. Bohr's model was first introduced in 1913. This model of the atom depicts a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus much like the planets travel around the Sun, but the electrostatic forces product attraction instead of gravity. The model's key success was in explaining the Rydberg formula for the spectral emission lines of atomic hydrogen. It is, basically, a modification of the Rutherford model used for quantum physics purposes.
The Bohr model was an improvement on older atomic models, but it too has been rendered obsolete by ongoing scientific research. Although considered to be obsolete, it is still taught as an introduction to quantum mechanics and in early secondary school science classes. Once students are advanced enough in their comprehension, they are introduced to the more accurate valence shell atom. At some time in the future this model of the atom may be proven to be too rigid in its scope.
The Bohr model built on the Rutherford theory. Rutherford proposed that electrons orbited the nucleus much like a planet around the Sun. The drawback to the theory was that based on his theory, electrons would be emitting(losing) their charge and spiral into the nucleus, making all atoms unstable. Bohr proposed several changes to that model: electrons can only travel in special orbits at a certain set of distances from the nucleus with specific energies, electrons do not continuously lose energy as they travel. They can only gain and lose energy by jumping from one allowed orbit to another, absorbing or emitting electromagnetic radiation with a frequency determined by the energy difference of the levels according to the Planck relation, and that the frequency of the radiation emitted at an orbit is the reciprocal of the classical orbit period. This model is restricted in a few ways, but does allow for classical mechanics to explain many things while having an allowance for quantum rules.
The Bohr model begins to run into problems with heavier atoms. Other shortcomings of the model are:gives an incorrect value for the ground state orbital angular momentum, fails to explain much of the spectra of larger atoms, and the model also violates the uncertainty principle because it considers electrons to have known orbits and definite a radius. These two things can not be directly known at the same time.
Here is a good ink
about the Bohr atom model
. Here on Universe Today we have a couple of great articles on the topic: one is about the Bohr model and the other is
about Dr. Bohr himself
. Astronomy Cast offers a good episode
about how molecules behave in space
.
 Universe Today
Universe Today