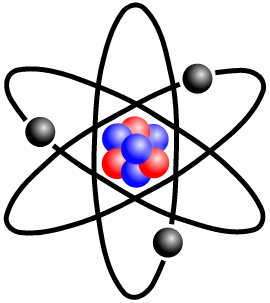
[/caption]The atom definition is: A unit of matter, the smallest unit of an element, having all the characteristics of that element and consisting of a dense, central, positively charged nucleus surrounded by a system of electrons. The entire structure has an approximate diameter of 10
-8
centimeters and characteristically remains undivided in chemical reactions except for limited removal, transfer, or exchange of certain electrons. Essentially, it is the smallest possible part of an element that still remains the element.
Under normal circumstances an atom can be broken down into any smaller particles, but we humans, have devised ways to break the atom apart. That is the entire basis of the atom bomb, particle colliders, and quarks. It takes high speed, high energy smashing of particles to break an atom. A particle collider does that and helps us understand some of the theories in particle physics. The results of an atom bomb are widely known. Quarks are extremely small particles that do not exist alone. They must group to form the protons, electron, and neutrons normally found in a single atom of an element. They have only been found as a result of a particle collider and in theory.
An atom itself is made up of three tiny kinds of particles called subatomic particles: protons, neutrons, and electrons. The protons and the neutrons make up the center of the atom called the nucleus and the electrons fly around above the nucleus in a small cloud. The electrons carry a negative charge and the protons carry a positive charge. In a normal (neutral) atom the number of protons and the number of electrons are equal. Often, but not always, the number of neutrons is the same, too. The opposing forces(negative and positive) attract and repel each other in the right way so as to keep the atom together. The universe could be looked at as one large atom. Everything in space attracts and repels just right so as to keep the whole together.
One type of theoretical ion propulsion spacecraft would have to take advantage of this atomic attraction and repulsion to operate. It takes advantage of magnetism and electricity to push a ship through space. Electricity, generated by the ship's solar panels, gives a positive electrical charge to atoms inside the chamber. They are pulled by magnetism towards the back of the ship and then pushed by magnetic repulsion out of the ship. This steady stream of atoms going out of the spacecraft gives it the thrust it needs to go forward through space. NASA has tested other types of ion propulsion and found them lacking.
Here is
another atom definition
. Here on Universe Today we have a great article
about atoms.
Astronomy Cast has a good question and answer episode
about interstellar travel
, including a NASA link about ion propulsion.
Source:
Wikipedia
 Universe Today
Universe Today