[/caption]
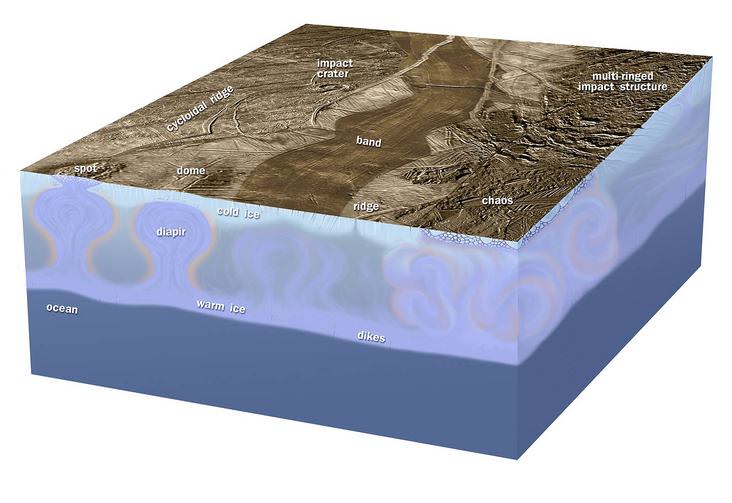
The more we explore our solar system, the more we find things in common. Jupiter’s frigid moon – Europa – is about the size of our satellite and – like Earth – home to some very hostile environments. Underneath what is surmised to be an icy crust a few miles deep, Europa may possess an acidic ocean that could extend down as much as 100 miles (160 km) below the surface. We know from exploring our home planet that life happens under some very extreme conditions here… But what about Europa? What are the chances that life could exist there, too?
Check out liquid water on Earth and you’ll find some form of life. As a given, scientists hypothesize other worlds which contain water should also support life. According to recent studies, Europa’s ocean might even be saturated with oxygen – further supporting these theories. However, there’s a catch. Like Earth, surface chemicals are continually drawn downward. According to researcher Matthew Pasek, an astrobiologist at the University of South Florida, this could constitute a highly acidic ocean which “is probably not friendly to life — it ends up messing with things like membrane development, and it could be hard building the large-scale organic polymers.”
According to Charles Choi of Astrobiology Magazine, “The compounds in question are oxidants, which are capable of receiving electrons from other compounds. These are usually rare in the solar system because of the abundance of chemicals known as reductants such as hydrogen and carbon, which react quickly with oxidants to form oxides such as water and carbon dioxide. Europa happens to be rich in strong oxidants such as oxygen and hydrogen peroxide which are created by the irradiation of its icy crust by high-energy particles from Jupiter.”
Although it’s speculation, if Europa produces oxidants, they may also be drawn toward its core from ocean motion. However, it might be infused with sulfides and other compounds creating sulfuric and other acids before supporting life. According to the researchers, if this has happened for just half of Europa’s lifetime, the result would be corrosive, with a pH of about 2.6, “about the same as your average soft drink,” Pasek said. While this wouldn’t prohibit life from forming, it wouldn’t make it easy. Emerging life forms would have to be quick to consume oxidants and build an acid tolerance – a process which could take as much as 50 million years.
Are there similar acid-lovin’ lifeforms on Earth? You bet. They exist in acid mine drainage found in Spain’s Rio Tinto river and they feed on iron and sulfide for their metabolic energy. “The microbes there have figured out ways of fighting their acidic environment,” Pasek said. “If life did that on Europa, Ganymede, and maybe even Mars, that might have been quite advantageous.” It is also possible that sediments at the bottom of Europa’s ocean may neutralize the acids, even though Pasek speculates this isn’t likely. One thing we do know about an acidic ocean is that it dissolves calcium-based materials such as bones and shells.
It’s a lesson repeated on Earth…
Right now our oceans are absorbing excess carbon dioxide from the air which – when combined with seawater – forms carbonic acid. While it is mostly neutralized by fossil carbonate shells at the ocean’s bed, if it’s absorbed too quickly it can have some major ramifications on sea life such as coral reefs, plankton and mollusks. According to a recent study, this acidification is happening faster (thanks to human carbon emissions) than it has during four major extinction events on Earth in the last 300 million years.
“What we’re doing today really stands out,” said lead author Bärbel Hönisch, a paleoceanographer at Columbia University’s Lamont-Doherty Earth Observatory. “We know that life during past ocean acidification events was not wiped out—new species evolved to replace those that died off. But if industrial carbon emissions continue at the current pace, we may lose organisms we care about—coral reefs, oysters, salmon.”
According to this new research, our carbon dioxide levels have escalated by 30% in the last century. This means we’ve jumped to to 393 parts per million, and ocean pH has fallen by 0.1 unit, to 8.1–an acidification rate at least 10 times faster than 56 million years ago, says Hönisch. If this continues, the Intergovernmental Panel on Climate Change predicts the pH may drop as much as another 0.3 units… a drop that will constitute major biologic changes. While you might scoff at the extinction of a few forms of plankton or the annihilation of a small coral or shellfish, there is a ripple effect that cannot be denied.
“It’s not a problem that can be quickly reversed,” said Christopher Langdon, a biological oceanographer at the University of Miami who co-authored the study on Papua New Guinea reefs. “Once a species goes extinct it’s gone forever. We’re playing a very dangerous game.”
It may take decades before ocean acidification’s effect on marine life shows itself. Until then, the past is a good way to foresee the future, says Richard Feely, an oceanographer at the National Oceanic and Atmospheric Administration who was not involved in the study. “These studies give you a sense of the timing involved in past ocean acidification events—they did not happen quickly,” he said. “The decisions we make over the next few decades could have significant implications on a geologic timescale.”
For now, we’ll look to Europa and wonder at what may exist below its frozen waves. Is there an acid-loving form of life just waiting to bubble to the surface for us to find? Right now researchers are developing a drill which could assist in looking for extreme forms of life. The “penetrator” could eventually be part of a Europa exploration mission which could begin as early as 2020.
“Penetrators are the most feasible, cheapest and safest option for a landing on Europa today, and the knowledge to build those is there,” said Peter Weiss, a post-doc now at the National Center for Scientific Research (CNRS) in France. “Otherwise, we won’t have any confirmation on astrobiology on Europa — or maybe even in the solar system — during our lifetime.”
Original Story Source: Astrobiology Magazine. For Further Reading: Physorg.com.