When researchers look up at the sky and wonder if we’re not alone, they also realize the origins of life here on Earth might hold the key to finding out. The chaotic chemical soup of our early world eventually led to the staggering complexity of modern life, but how exactly did it start? Proteins were one of the key ingredients in the early years, but we’re still only just discovering how these marvels of modern biology first managed to fold, function, and survive. A new review paper, The borderlands of foldability: lessons from simplified proteins, published recently in Trends in Chemistry, showcases how scientists are attempting to answer this question - by researching “simplified proteins”.
Modern proteins are notoriously complex. Each is composed of some combination of one of twenty distinct amino acids. But it’s highly unlikely that early Earth had access to all twenty of those building blocks of proteins. The earliest peptides were likely short, simple, and composed only of peptides that were either created natively in the environment or produced by life with a very primitive metabolism. Granted, we can’t exactly dig up ancient fossils of early proteins or amino acids to prove this point, but that seems like a reasonable assumption to make.
To simulate this simplified environment, scientists perform what they call “alphabet reduction” by rebuilding proteins with restricted alphabets of only 7 to 14 amino acids. And what they manage to build with such a limited toolkit is still amazing. Scientists have built proteins that will successfully fold into 3D structures while completely excluding entire classes of complex building blocks like simple or aromatic amino acids.
Fraser discusses how we found the build blocks of life in OSIRIS-REx’s samplesThis proves that the core architectures of proteins needed to produce life require startlingly little information. A “prebiotic” alphabet of roughly ten amino acids is more than enough to get the ball rolling on more complex life forms. One of the most famous ideas in this space was developed back in 1966 by Richard Eck and Margaret Dayhoff, who hypothesized that ancient, symmetric proteins were formed by the duplication and fusion of short, simple peptides. Modern science has more or less proven that idea in practice, with scientists watching sets of simple peptides “homo-oligomerize” - essentially snapping together like Lego bricks to form symmetric, fully functional proteins.
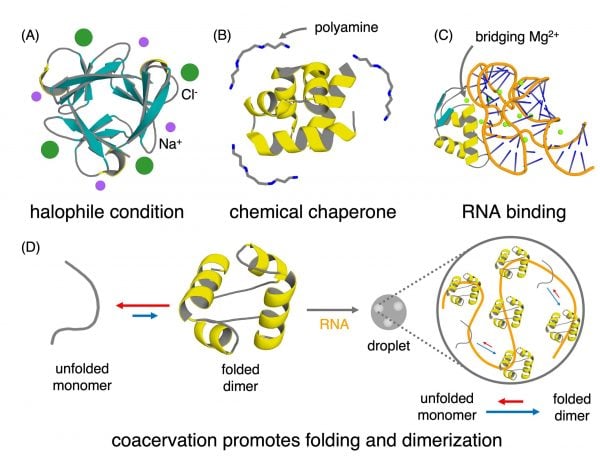
But back on early Earth, these proteins would not have formed in a vacuum. The environment itself might have played an active role in supporting them. Despite its harshness, the environment of the early Earth actually acted like scaffolding to help support early simple proteins that were only “marginally stable” by themselves. Several factors contributed, including hypersaline oceans, where high salt concentrations exerted external effects on protein stability, such as by “charge screening” that can force proteins to fold.
Another potential environmental factor was the presence of compounds like polyamines and dications (i.e. an atom that carries at +2 charge, like magnesium) that could have acted like molecular glue. Finally, the crowded environment inside a coacervate - the concentrated chemical droplets that separated early life from its environment before cellular membranes were developed - could have promoted both peptide folding and oligomerization that wouldn’t have worked in more isolated environments.
Lecture on the search for life in Enceladus. Credit - SVAstronomyLectures YouTube ChannelBut perhaps the most exciting part about this research area lately has been the introduction of AI. AlphaFold is a powerful AI tool capable of predicting the structure of proteins and analyzing their folding. But more importantly from a space perspective, these large language models of protein libraries allow us to simulate what might have been happening both billions of years ago on Earth, but also actively happening on Enceladus or Europa.
The jump between an inert world with only a chemical soup to the living, breathing biosphere we have today seems massive. But, like most other things in life, it was created step by step. Early, prebiotic protein folding was one of those critical steps, and studying how they did so will be key to understanding that link in the chain. Relying on simple, repeating chemical fragments of a restricted number of abundant chemicals supported by a harsh environment seems a difficult place to start. But it seems to be exactly where we did - and it's a good idea to keep that in mind as we search for other early stage life journeys throughout the cosmos.
Learn More:
ELSI - The hidden language of life’s early proteins
K. Seya et al - The borderlands of foldability: lessons from simplified proteins
UT - Scientists Make a Game-Changing Find in the Bennu Asteroid
UT - All Life on Earth is Made up of the Same 20 Amino Acids. Scientist Now Think They Know Why
 Universe Today
Universe Today