Life on Earth depends on organic chemicals—the elements carbon, hydrogen, nitrogen, oxygen, phosphorous, and sulfur—often referred to as CHNOPS. These elements enable life and make up about 98% of all living matter. But they don't do it alone. Biochemical activity inside of cells also requires small amounts of metals. These metals enable critical biochemical reactions.
To understand how life on Earth evolved, scientists work to understand the availability of CHNOPs and bio-important metals and how they cycle through the environment.
Molybdenum (Mo) is an important metal in biochemistry and lends speed to reactions in cells. These faster reactions make complex life possible. Mo is an essential element in most lifeforms, and is a component of their critical enzymes. While scientists have known about the role it plays for a long time, the history of its availability and how it shaped life on the planet hasn't been so clear. New NASA-funded research has uncovered molybdenum's history in the biosphere and how life relied on it long ago.
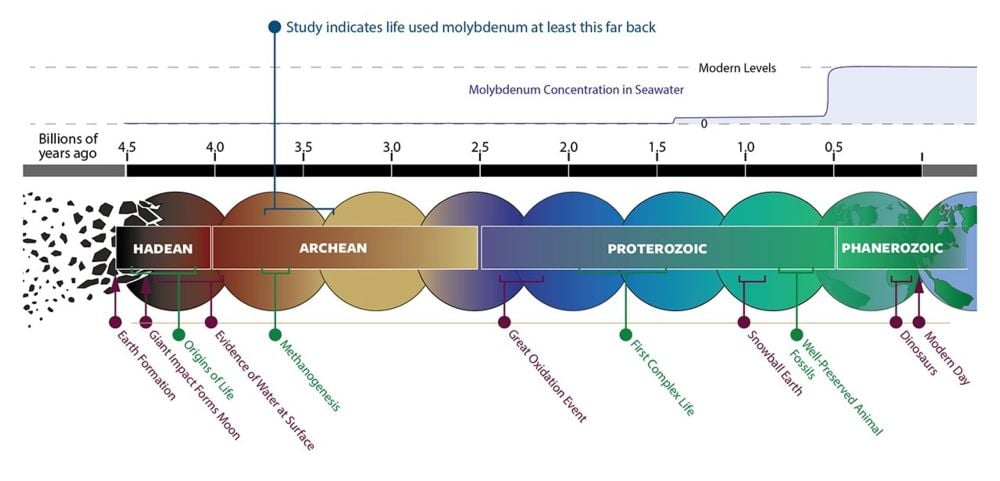
The availability of elements changed as Earth evolved and as chemical cycles changed. Previous research showed that Mo was scarce in Earth's early periods, and that its scarcity was a barrier to its use by lifeforms. But the new work shows that life relied on Mo, even when it was present in smaller amounts.
The research is "Biological use of molybdenum and tungsten stems back to 3.4 billion years ago," and it's published in Nature Communications. The lead author is Aya Klos from the Department of Bacteriology at the University of Wisconsin-Madison. The senior author is Betül Kaçar, head of the Kaçar Lab at the University of Wisconsin-Madison. Kaçar also leads MUSE, a NASA Interdisciplinary Consortia for Astrobiology Research (ICAR) at UW-Madison.
“Molybdenum sits at the catalytic center of enzymes that run major carbon, nitrogen, and sulfur reactions,” explained Kaçar in a press release. “Asking when life began using molybdenum is really asking when some of the most consequential metabolic strategies became possible,” said Kaçar.
Another metal, Tungsten (W), plays a similar role as Mo does, and some modern-day organisms in extreme environments rely on it instead of Mo. Scientists have wondered if organisms began using W and then evolved to use Mo as its availability increased. But this research says otherwise.
The researchers tracked Mo's abundance throughout Earth's history and its use by organisms across the branches of the tree of life. They found that despite molybdenum's ancient scarcity, ancient microbes still made use of it. They found that the same is true of W, and that it's unlikely that organisms relied first on W and then on Mo.
“Our work shows that both molybdenum and tungsten-using enzyme systems have Archean roots, which suggests that early life likely worked with both metals rather than following a simple “tungsten first, molybdenum later” story,” said Kaçar. “We argue that molybdenum use is far older than many models assumed, with molecular dating placing molybdenum utilization back into the Eoarchean to Mesoarchean, roughly 3.7–3.1 billion years ago, well before the Great Oxidation Event.”
Molybdenum wasn't widely available on the early Earth, but it was available in some locales. Hydrothermal vents were early sources for Mo and other metals like iron, zinc, manganese, and tungsten. (That's one of the reasons that astrobiologists wonder about similar vents on other ocean worlds and our Solar Systems's ocean moons.) So even though Mo wasn't widespread, its localized availability was critical.
“Even if Archean seawater held little dissolved molybdenum overall, localized systems such as hydrothermal vents could still have supplied usable amounts of molybdenum and other metals,” said Kaçar.
The critical part of this is that even though multiple metals were available in these locales, life utilized Mo. Mo is like the Swiss Army Knife of metals when it comes to cellular functions. So despite its overall scarcity, and despite the availability of other metals in the same locales, Mo became central.
“Molybdenum may have been worth “choosing” because it enables catalysis across a broad range of substrates and redox conditions,” said Kaçar. “In other words, scarcity did not make molybdenum unimportant; its catalytic advantages may have made it worth evolving ways to acquire and use.”
In their work, the researchers explain that Mo and enzymes based on it became more widespread before and during the Great Oxygenation Event, a critical period in Earth's history. "Intriguingly, the emergence and diversification of molybdoenzymes preceding and during the GOE, when oxidative weathering began delivering increased amounts of Mo from land to the ocean, suggests that Mo-based biochemistry may have partly evolved in response to a shift in Mo supply, i.e., from hydrothermal vents to riverine input," they write.
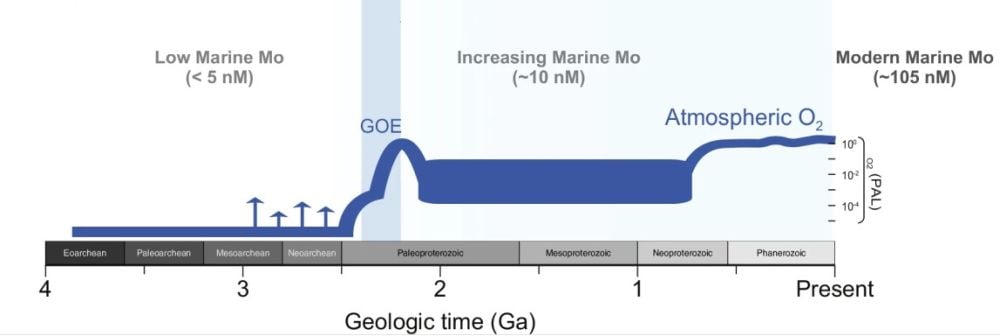 This figure illustrates how Molybdenum became more available in seawater before and during the GOE. Image Credit: Klos et al. 2026. NatComm.
This figure illustrates how Molybdenum became more available in seawater before and during the GOE. Image Credit: Klos et al. 2026. NatComm.
Our search for life is centered on biosignatures, certain chemicals and molecules in atmospheres. But as scientists learn more about how life used elements in its evolution, even scarce ones like Mo, it could signal a change in how they approach astrobiology.
“Our NASA ICAR shows that mapping the evolutionary history of bio-essential elements on Earth can help us predict what life on other worlds might use, and that different abiotic inventories could lead to different biological element choices,” said Kaçar. “Life detection should be metal-aware, redox-aware, and evolution-aware. We should look not just for ‘Earth-like life now,’ but for biochemical strategies that would make sense on a planet with a different history of oxygenation and metal availability.”
Earth's transition from an ancient, raw chemical soup to the planet we inhabit now, one resplendent with life, is a long, intricate story. We may never know exactly how life first started, but scientists have shown that they can trace life's evolution in minute detail. As their understanding of how life used scarce elements like Mo to grow in efficiency and complexity, they're learning not only about Earth life, but what other habitable exoplanets might go through.
It can only help in our search for life.
 Universe Today
Universe Today