The existence of water on Mars is a contentious subject. We know there used to be water on the surface of the planet, though it's long gone now. We know there's frozen water underground in the world, and we know there's water vapour in the air. But life needs liquid water.
Could there be liquid water on Mars?
A new study shows how salty water could emerge from the atmosphere onto Mars' surface under the right conditions.
The research is based on hygroscopic salts. They have an affinity for water and can absorb it from the atmosphere. The process is called deliquescence, and while previous research and lab experiments show deliquescence takes place on Mars, there are gaps in our understanding of how, when, and where it might happen.
This new paper helps to fill in some of the gaps.
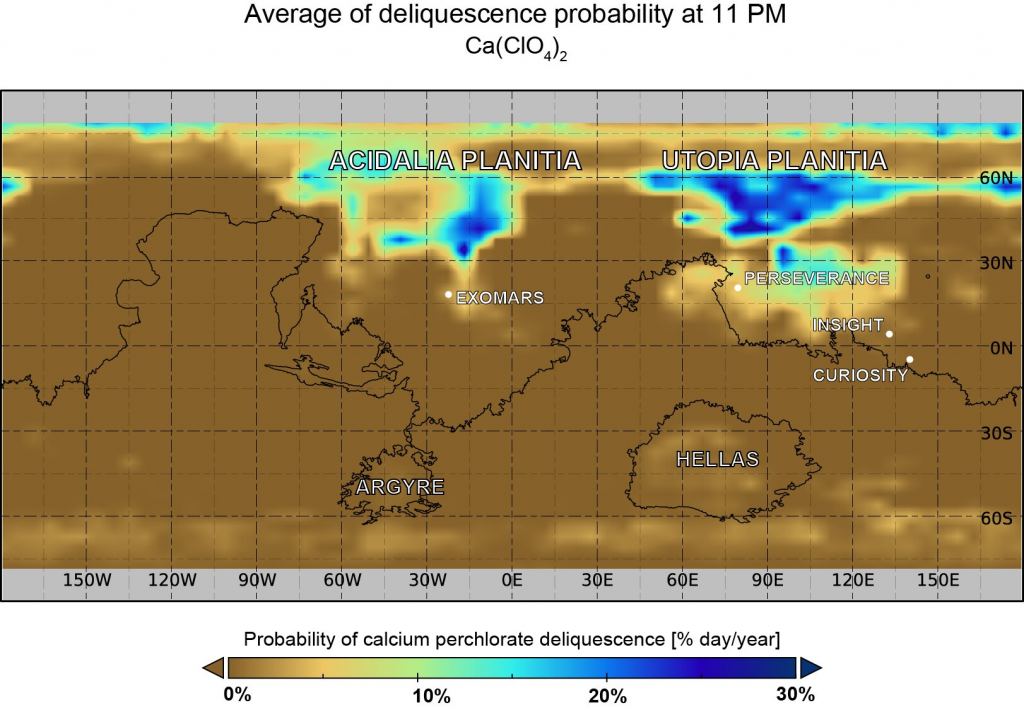
The paper's title is " Deliquescence probability maps of Mars and key limiting factors using GCM model calculations." The authors are Bernadett Pál and Ákos Kereszturi, planetary scientists at Konkoly Observatory in Budapest, Hungary. The paper is published in the journal Icarus.
Liquid water faces some hurdles on Mars. There's frozen water, but it doesn't form liquid water if it thaws. The temperature and the atmospheric pressure are so low that water sublimates from ice to vapour. Stable fresh liquid water can't form on Mars' surface.
But salty water remains liquid at much lower temperatures than freshwater.
Calcium perchlorate is a hygroscopic salt that's common on Mars. MSL Curiosity found it there, and so did the Phoenix Lander. Research shows that calcium perchlorate accounts for up to 1% of Martian soil by weight.
Calcium perchlorate can both absorb and adsorb water. In chemistry, absorption means the diffusion of particles of gas or liquid into liquid or solid materials. Adsorption is different. Adsorption is the adherence of particles to the surface of the adsorbent. Deliquescence is when, under the right conditions, a hygroscopic salt absorbs and adsorbs enough water to form a brine.
Deliquescence is a well-studied phenomenon here on Earth, where it's important in everything from agriculture to food science. But we have no in-situ knowledge of how it works on Mars. Our understanding of deliquescence on Mars is based on powerful computer simulations.
The authors modelled two salts in this study: calcium perchlorate and magnesium perchlorate. Brines made of both of these salts can remain liquid down to -70C (-90F, 203 K) as long as the relative humidity is high enough.
Their models covered an entire Martian year and the planet's entire surface. The authors found that both hemispheres have conditions that promote calcium perchlorate deliquescence. But ideal conditions are between local spring through late summer. Inside that, the best conditions are from late night until early morning.
The study shows that the entire planetary surface likely doesn't support calcium perchlorate deliquescence. But most regions north of 30°N latitude can be ideal for brine emergence late at night, while in the early morning hours, the large basins (Acidalia Planitia, Utopia Planitia) are promising.
If there's any extant life on Mars, it likely needs water. If Mars has a water cycle, it's focused on brines. Understanding deliquescence and the formation of brines can only help scientists find any of that life if it exists.
Dr. Edgard Rivera-Valentín is a NASA early career fellow at USRA Lunar and Planet Institute. Dr. Rivera-Valentin studies habitability and brine formation in particular. He's not involved in this research but has authored and co-authored multiple research papers and white papers on habitability and Mars' salty water cycle. In a press release, Dr. Rivera-Valentin commented on this new research.
"The recent work by Ms. Pál and Dr. Kereszturi provides important insights into the potential to form brines on Mars, which is very relevant to understanding Mars' biologic potential," he said. "Additionally, vetting expectations of where and when brines can form and show that such conclusions are independent of the type of climate model used, like was done here, by comparing to my own work published in 2020, makes these findings robust. Their detailed work looking at seasonality and diurnal effects furthers the field by providing important information for future missions to Mars and their experiments, like the BOTTLE experiment onboard the ExoMars Rosalind Franklin rover" he added.
Study authors Pál & Kereszturi are looking ahead to future missions to Mars, too. The BOTTLE (Brine Observation Transition To Liquid Experiment) is part of the ExoMars 2022 mission. BOTTLE will monitor in-situ, during one full Martian year, the diurnal and seasonal changes of the electric conductivity of four salts found on Mars: calcium chloride (CaCl 2); ferric sulphate Fe 2 (SO 4) 3; magnesium perchlorate Mg(ClO 4 )2 and sodium perchlorate (NaClO 4). Part of BOTTLE's mission is to serve as an In-Situ Resource Utilization demonstration for an atmospheric water-capturing system for human exploration.
The HABIT (HabitAbility, Brine Irradiation and Temperature) instrument will perform the BOTTLE experiment.
"I can't wait for the first results of the ExoMars BOTTLE Experiment. I've been working on modelling the possibilities for liquid water formation on the Martian surface for years, and we will be able to learn a great deal from the first true experimental results there," Ms. Pál said.
Oxia Planum is ExoMars' planned landing site. In the study's simulations, Oxia Planum showed ideal conditions for liquid brine formation in both late night and early morning hours. So BOTTLE's results should be robust.
When Recurring Slope Linae (RSL) were first spotted on Mars, they were a puzzle. The Compact Reconnaissance Imaging Spectrometer for Mars (CRISM) instrument aboard NASA's Mars Reconnaissance Orbiter detected magnesium perchlorate, magnesium chlorate and sodium perchlorate within recurring slope lineae. Now it seems like RSL may be a foundational part of Mars' salty water cycle. But is there any potential for Mars' brines to support life?
Studies of extremophiles grown in perchlorate and chlorate solutions suggest that microbes could survive in Mars' brines. A group of scientists led by Mark Schneegurt, a professor of biological sciences at Wichita State University in Kansas, USA, found that several species of halotolerant, i.e. salt-tolerant bacteria, were able to grow in high concentrations of chlorate salts.
Finding water on Mars is about two things: the search for life and human missions to the planet. In their paper's conclusion, the authors write, "As the search for extraterrestrial habitability (past or present) and even future crewed mission plans (requiring in-situ water production) are gaining popularity, the importance of searching and predicting liquid water formation and the stability of solutions on the surface of Mars is increasing."
"Our results suggest that there is a good chance for calcium perchlorate deliquescence across larger regions on Mars, and the possibly ideal times are spread across several hours," they explain. "Our deliquescence probability maps could be used as a good startings point in finding interesting locations for local ephemeral brine studies or as a preliminary guide in planning future, liquid brine-related in-situ missions."
 Universe Today
Universe Today