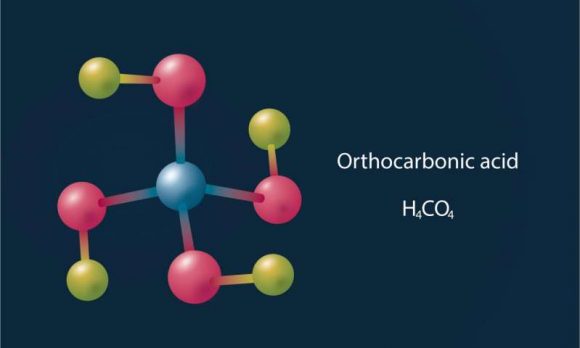
“Hitler’s acid” is a colloquial name used to refer to Orthocarbonic acid – a name which was inspired from the fact that the molecule’s appearance resembles a swastika. As chemical compounds go, it is quite exotic, and chemists are still not sure how to create it under laboratory conditions.
But it just so happens that this acid could exist in the interiors of planets like Uranus and Neptune. According to a recent study from a team of Russian chemists, the conditions inside Uranus and Neptune could be ideal for creating exotic molecular and polymeric compounds, and keeping them under stable conditions.
The study was produced by researchers from the Moscow Institute of Physics and Technology (MIPT) and the Skolkovo Institute of Science and Technology (Skoltech). Titled “Novel Stable Compounds in the C-H-O Ternary System at High Pressure”, the paper describes how the high pressure environments inside planets could create compounds that exist nowhere else in the Solar System.
Professor Artem Oganov – a professor at Skoltech and the head of MIPT’s Computational Materials Discovery Lab – is the study’s lead author. Years back, he and a team of researchers developed the worlds most powerful algorithm for predicting the formation of crystal structures and chemical compounds under extreme conditions.
Known as the Universal Structure Predictor: Evolutionary Xtallography (UPSEX), scientists have since used this algorithm to predict the existence of substances that are considered impossible in classical chemistry, but which could exist where pressures and temperatures are high enough – i.e. the interior of a planet.
With the help of Gabriele Saleh, a postdoc member of MIPT and the co-author of the paper, the two decided to use the algorithm to study how the carbon-hydrogen-oxygen system would behave under high pressure. These elements are plentiful in our Solar System, and are the basis of organic chemistry.
Until now, it has not been clear how these elements behave when subjected to extremes of temperature and pressure. What they found was that under these types of extreme conditions, which are the norm inside gas giants, these elements form some truly exotic compounds.

As Prof. Oganov explained in a MIPT press release:
“The smaller gas giants – Uranus and Neptune – consist largely of carbon, hydrogen and oxygen. We have found that at a pressure of several million atmospheres unexpected compounds should form in their interiors. The cores of these planets may largely consist of these exotic materials.”
Under normal pressure – i.e. what we experience here on Earth (100 kPa) – any carbon, hydrogen or oxygen compounds (with the exception of methane, water and CO²) are unstable. But at pressures in the range 1 to 400 GPa (10,000 to 4 million times Earth normal), they become stable enough to form several new substances.
These include carbonic acid, orthocarbonic acid (Hitler’s acid) and other rare compounds. This was a very unusual find, considering that these chemicals are unstable under normal pressure conditions. In carbonic acid’s case, it can only remain stable when kept at very low temperatures in a vacuum.

At pressures of 314 GPa, they determined that carbonic acid (H²CO³) would react with water to form orthocarbonic acid (H4CO4). This acid is also extremely unstable, and so far, scientists have not yet been able to produce it in a laboratory environment.
This research is of considerable importance when it comes to modelling the interior of planets like Uranus and Neptune. Like all gas giants, the structure and composition of their interiors have remained the subject of speculation due to their inaccessible nature. But it could also have implications in the search for life beyond Earth.
According to Oganov and Saleh, the interiors of many moons that orbit gas giants (like Europa, Ganymede and Enceladus) also experience these types of pressure conditions. Knowing that these kinds of exotic compounds could exist in their interiors is likely to change what scientist’s think is going on under their icy surfaces.
“It was previously thought that the oceans in these satellites are in direct contact with the rocky core and a chemical reaction took place between them,” said Oganov. “Our study shows that the core should be ‘wrapped’ in a layer of crystallized carbonic acid, which means that a reaction between the core and the ocean would be impossible.”

For some time, scientists have understood that at high temperatures and pressures, the properties of matter change pretty drastically. And while here on Earth, atmospheric pressure and temperatures are quite stable (just the way we like them!), the situation in the outer Solar System is much different.
By modelling what can occur under these conditions, and knowing what chemical buildings blocks are involved, we could be able to determine with a fair degree of confidence what the interior’s of inaccessible bodies are like. This will give us something to work with when the day comes (hopefully soon) that we can investigate them directly.
Who knows? In the coming years, a mission to Europa may find that the core-mantle boundary is not a habitable environment after all. Rather than a watery environment kept warm by hydrothermal activity, it might instead by a thick layer of chemical soup.
Then again, we may find that the interaction of these chemicals with geothermal energy could produce organic life that is even more exotic!
Further Reading: MIPT, Nature Scientific Reports


Using the term ‘Hitler’s acid’ in a serious article on science is ludicrous, inappropriate and offensive. The swastika was used by various cultures for millenia before being misappropriated in reflected form by the twisted Nazi thugs. The oldest swastikas are neolithic. The swastika is a sacred and auspicious symbol in Buddhism, Hinduism, and Jainism. How about taking a name from one of those traditions or just calling it ‘swastik acid’? Why honour the most notorious war criminal in human history by naming a chemical compound after him? That is in very bad taste.
Rob Stuart
Utrecht, NL
@robst247- What is ludicrous is your obvious lack of research into the term prior to formulating and posting your “social sensitivity” opinion. In addition to it looking like a swastika Orthocarbonic acid is considered highly unstable. How exactly do you rationalize a reference to being unstable as a means of honoring Hitler. In science a comment or an opinion without research is called ignorance.
How do you know, ‘theleadernews’, whether I did prior research or not? That’s just your knee-jerk assumption, and therefore meaningless. The article itself refers to the instability of the acid compound; therefore, your reaction is beside the point. If Hitler had been a chemist and had discovered this compound, it would have been appropriate to name it after him. He did not; therefore, using his name in this context is inappropriate. The acid is not Hitler’s, neither is the swastika. That narcissistic, sexually dysfunctional failure and his demented cronies ignorantly misappropriated and forever besmirched a beautiful religious symbol.
Your objection is overruled. I rest my case.
robst247 correctly states that the swastika pre-dates National Socialism by millenia, but in doing so ignores that in modern times that symbol has been corrupted and embraced by those who wish hate & destruction upon their fellow man, even to present day. It is the argument that the Confederate battle flag is “nothing more than an historical artifact” raised to a higher power, and in being made likewise ignores current reality in favor of one wished for.
theleadernews correctly states that orthocarbonic acid is legitimately nick-named “Hitler’s acid,” and for legitimate reasons, but they too miss reality: anything so named in the scientific realm is considered “an honor,” no matter the properties of the item (e.g. “curium” & most of the other actinides/lanthanides). It is rare for anyone to actually refer to the chemical in this way outside of colloquial discussions among lab technicians for this very reason; the author’s choice to use the name in the headline is clearly intended as click-bait & nothing more.
Every article w/the Austrian Napoleon’s name in it is NOT a deliberate assault on everyone’s sensibilities, nor does it beg a flaming rebuttal of his Mustachioed Madness. Likewise, neither does the correct recitation of historical fact obviate the need for sensitivity to current events & conditions, regardless of what we all might wish for… in this case, a time when Nazi horror wasn’t held synonymous with an otherwise benign “good-luck charm.”
@Smokey – Your statement is completely FALSE: “anything so named in the scientific realm is considered “an honor,” no matter the properties of the item” Absolutely not my friend. Some chemicals are named to honor, but the MAJORITY ARE given names NOT in honor but as description to their characteristic nature. Here are two that have negative meanings that have ABSOLUTELY NOTHING to do with honor.
1) Cobalt – From German Kobold, which means “evil spirit”. The metal was named by miners, because it was poisonous and troublesome (polluted and degraded other mined elements).
2) Osmium – From Greek (osme), meaning “a smell”
theleadernews persists in being self-righteous to the point of being combative, by focusing on one particular word used, rather than all of what was written.
“Anything so named” means anything named for a particular person, as those with a degree of reading comprehension will understand instantly, and as the elements which I specifically referenced demonstrate. As I pointed out, the Curies are honored through such naming (an element, as well as a unit of measure). One could argue that naming a strongly radioactive element as well as a unit of radiation emission after a pair of scientists killed BY radiation is a fairly macabre way to honor them, yet an “honor” it is nevertheless.
So a dose of reading comprehension raises my point from “completely false” to “utterly valid,” and my point regarding the rare usage of the term “Hitler’s acid” — as well as that of automatically lashing out at those whose views disagree with their own — stands untouched by theleadernews’ diatribe.
Sorry, that is misleading. There are many elements which are named for people, there are many elements which are not named for people. The point is that those which are named for people are so named to honour the people for whom they are named. Curium for the Curies, Seaborgium for Seaborg, and so on. In other words, when science names an element after a person, it is intended to honour this person. This mentioning of the naming by some people of a chemical as “Hitler’s Acid”, contributes nothing to the article itself. It should be added that the fact that others do the same thing is not a reason for not criticising it here. I would criticise it anywhere. It is the sort of thing that you would expect of tabloid journalism.
Au contraire, Smokey. Far from ignoring the embracing and corrupting of the swastika “by those who wish hate & destruction upon their fellow man”, I highlighted it in my comment, and that is precisely why I criticized the author’s use of their insane leader’s name in a popular article on science. Nor did I argue that doing so constitutes “a deliberate assault on EVERYONE’s sensibilities”; I said it was “ludicrous, inappropriate and offensive”. I meant offensive to SOME.
You may well be correct in asserting that the author’s use of that infamous surname in this context was “clickbait”. It was certainly not in good taste, and that is my point.
robst247’s original comment contained the following question: “How about taking a name from one of those traditions or just calling it ‘swastik acid’?” The reason is precisely because it is most memorably & notably associated in current, modern times with the Butcher of Berlin, as I pointed out; that fact is unfortunate but unavoidable. Certainly it is not the author who cast the moniker originally, as it has been around these many decades now — Mr. Williams simply makes use of it to generate site traffic — so he cannot be blamed for its creation either.
Smokey, Mr Williams CAN be blamed for using the moniker to generate site traffic, and that’s exactly what I’m doing! It’s a cheap trick and it’s tasteless.
robst247’s opinion appears to reflect an unawareness of the fact that a myriad of other outlets beat Mr. Williams to that exact punch. They should therefore widen the scope of their vitriol to include every other media outlet discussing the story… which according to a quick Google search appears to be ALL of them.
Happy ranting.
No, what he does is make an assertion that is simply not true. That the symbol of National Socialism under Adolph Hitler was a black swastika, is irrelevant. At most, what can be asserted is that the shape of the molecule in two dimensions is similar to that of the swastika.
So your original suggestion is that something that is UNSTABLE be associated with Buddhism, Hinduism, and Jainism. That is a complete CONTRADICTION as to the nature of these religions. Unless your purpose is to insult these religions. When logic is a contradiction it can ONLY be viewed as ignorance. You are guilty as charged – ignorance.
To ‘the leadernews’: There is no contradiction. As you may know, impermanence is a key concept in Buddhist and Hindu teachings. Therefore, giving an unstable molecule a name derived from one of those traditions would seem quite appropriate.
Sad to say but the bottom line truth is simply that both @robst247 and @Smokey rebuttals were in fact based on a “prior to post” false assumption that scientifically named chemicals are SOLELY relegated towards a form of “honor”. While some chemicals are a form of honor(as I stated) to the scientist that discovered them or the region the MAJORITY have NOTHING to do with HONOR. Whether they realize they made a false assumption or not is up to them to resolve in their minds. Regardless if they accept their error or not is totally up to them. Either way a simple lookup of how chemicals are named clearly reveals that this assumption of honor is simply a falsehood. No where will anyone find any reference to Hitler’s Acid ever being considered a form of honor. Its a false belief.
I’m usually pretty sardonic about the editorial decisions here. I will say something I think is funny or sarcastic about terraforming, or whatever. And I feel guilty for trolling! Wow. This one I’m just going to SMH. WTF were you thinking. On another topic, the internals of giant planets are fascinating and mysterious. Looking forward to getting more info out of Juno. Someday maybe a similar probe of the outer planets. Hopefully we won’t kill off public interest by committing PR blunders.
They lost me at “Under normal pressure any carbon, hydrogen or oxygen compounds (with the exception of methane, water and CO²) are unstable.”
I may be unstable after consuming ethanol, but the ethanol itself does not spontaneously decompose no matter how long it sits in a bottle. If all organic compounds were unstable under normal conditions there would be no such thing as organic chemistry, or life itself.
Here’s a clue Matt: exothermic reactions are a necessary, but not sufficient, condition for instability.
You beat me to it, although I had to read through all the crap above to find it. 😉
“Under normal pressure – i.e. what we experience here on Earth (100 kPa) – any carbon, hydrogen or oxygen compounds (with the exception of methane, water and CO²) are unstable.”
Pardon? We’re all based on carbon, hydrogen and oxygen compounds, so how can we exist? Surely ethanol at least is stable, or do I need to drink that gin immediately?
The reason they are using the name of Hitler is because he is one of the first going out space, invited by the Draco, to destroy more than one planet, but all solar system.
“Hitler’s Acid” = click-bait.
It’s such a pity that the first comments about an article are not about the science IN the article and we’re sidetracked by B/S.
yep, although the ridiculous reactions of some of the above commenters do serve as an indicator that it wasn’t the greatest name choice to call it “hitler’s acid”.
There is actually no up or down in the universe. If one is standing at the intersection of equator and Greenwich meridian lines of latitude and longitude on one opposite side of the earth he/she will point at his feet as down. One standing on the opposite direction on the other side of the globe will point in the same direction but as up. So up and down is not fixed in the universe
Ahem.. If you had read the article you would have seen that Matt DID NOT come up with this name.. If you feel you need someone to “blame” , then the Moscow Institute of Physics and Technology is a more likely ‘target’ for any condemnations.
The gas giants are not the only places where rare or otherwise unknown elements may exist. Our moon, due to bombardment by comets and asteroids over the milenia no doubt contains molecules equally as ‘rare’. Mars, Mercury, Venus and a plethora of our solar systems moons will no doubt also provide suprises! There might even be compounds that have the potential for changing the course of human history… LET’S GO find them!