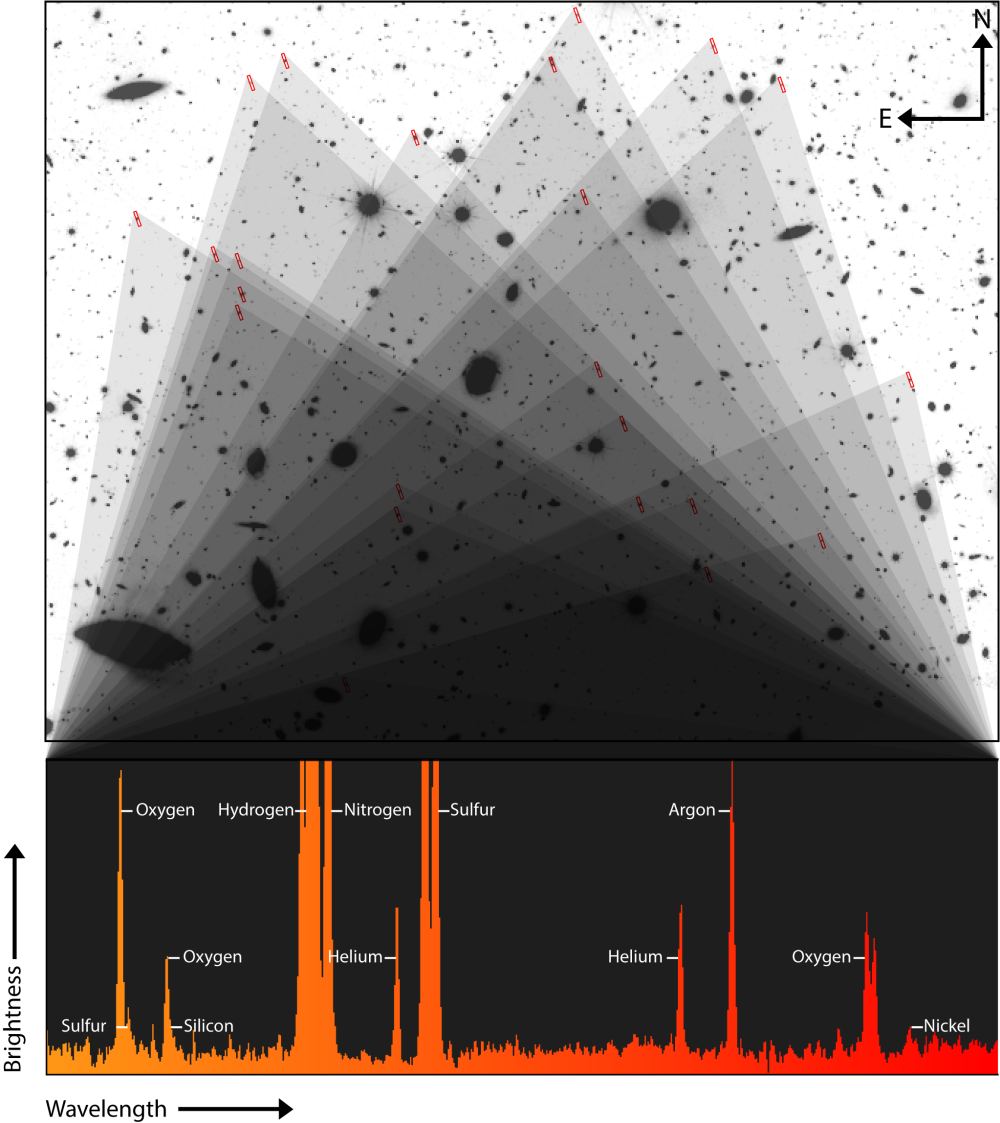
If the Universe has adolescent galaxies, they’re the ones that formed about 2 to 3 billion years after the Big Bang. New research based on the James Webb Space Telescope shows that these teenage galaxies are unusually hot. Not only that, but they contain some unexpected chemical elements. The most surprising element found in these galaxies is nickel.
Continue reading “Adolescent Galaxies are Incandescent and Contain Unexpected Elements”Adolescent Galaxies are Incandescent and Contain Unexpected Elements